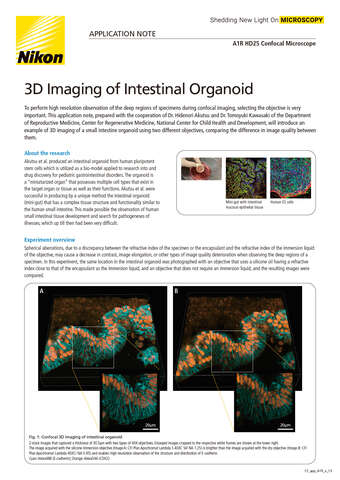
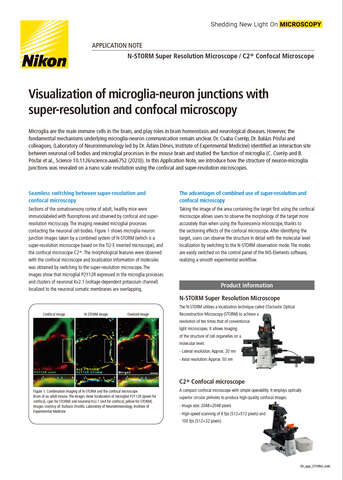
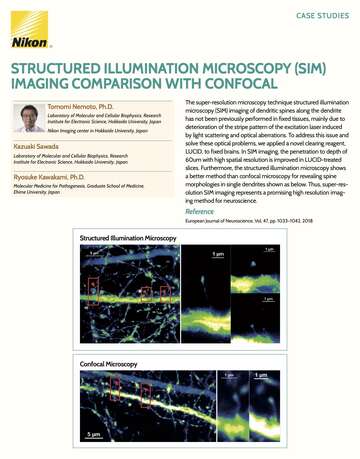
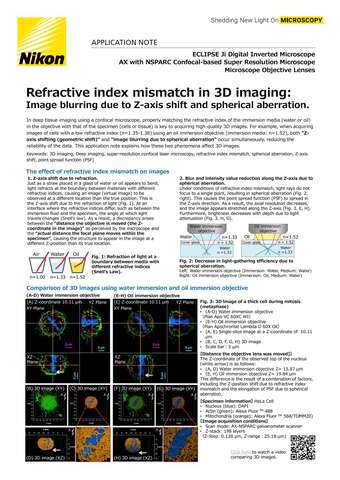
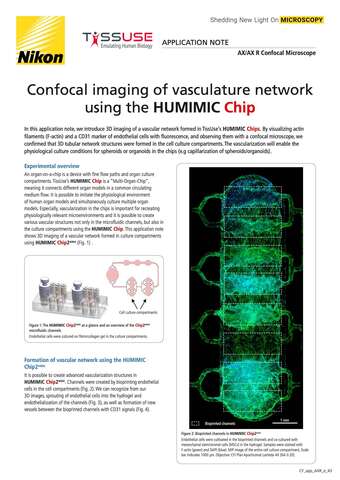
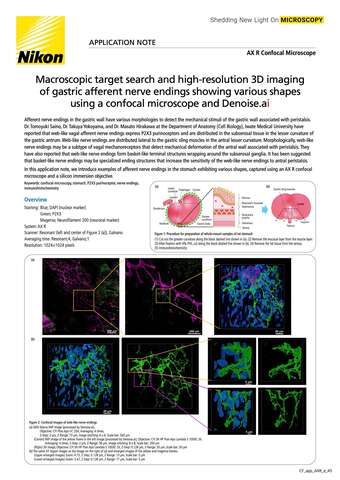
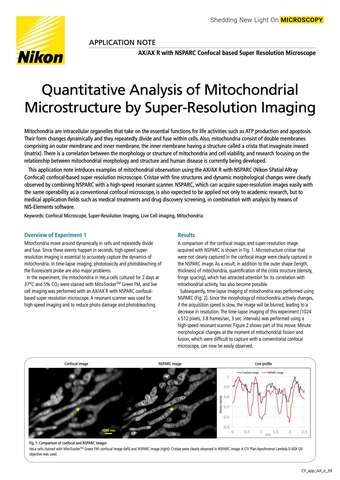
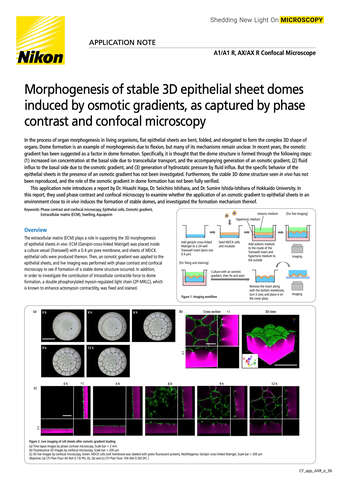
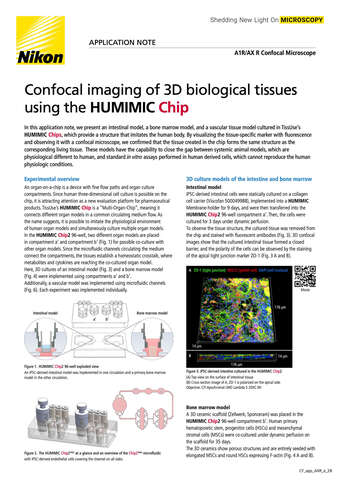
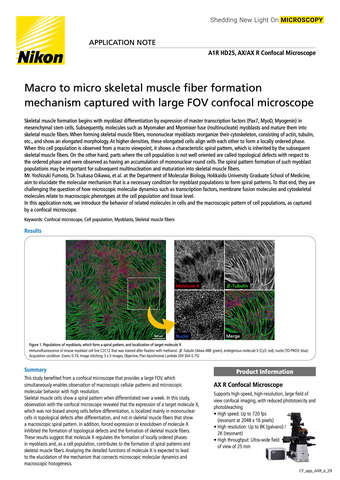
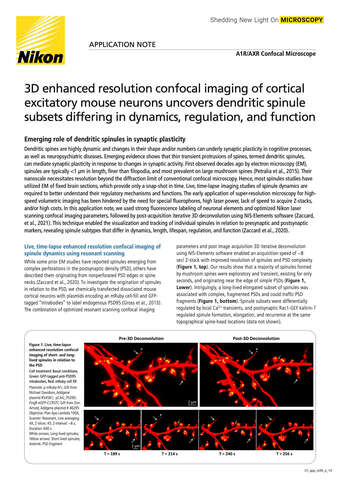
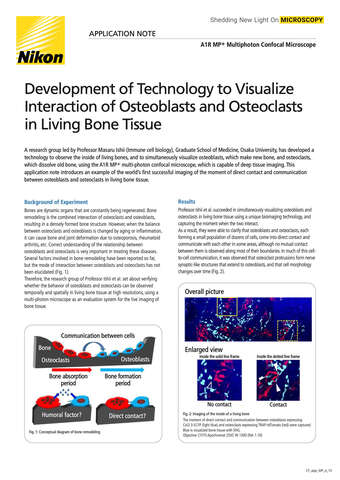
- ko Change Region
- Global Site
- 홈
- 제품 · 서비스
- 공초점 및 다광자 현미경
- AX / AX R with NSPARC
Glial cell surrounded by axons in a rat neuronal culture labeled for microtubules and actin
Dr. Christophe Leterrier, NeuroCyto, INP, Marseille, France
AX / AX R with NSPARC
공초점 현미경
애플리케이션 노트
애플리케이션 노트 필터링 기준