Alberto Diaspro, Ph.D.
Professor of Applied Physics, Department of Physics, University of Genoa
Director of the Department of Nanophysics, Istituto Italiano di Tecnologia
Équipement Nikon
- ECLIPSE Ti-E Inverted Microscope (see current model)
- A1R MP Multiphoton Confocal
- N-STORM Super-Resolution
Please tell us about your research.
My experience is in the design, realization and utilization of optical and biophysical instrumentation, as carried out with my research team. Our main aim is to improve on existing techniques to develop new, unique technologies. Our research centers on several main themes; one is the development of higher resolution imaging solutions, especially involving fluorescence. We are interested in techniques that give XY resolution below 30 nm and Z resolution below 100 nm. In order to do this we need to work with systems that have an excellent signal-to-noise ratio to preserve optical quality when imaging at nanometer resolution. We recently published a paper on this reporting about our IML-SPIM setup allowing 3D super-resolution on thick biological objects (Nature Methods 8, 1047–1049 (2011)).
Something else we’re interested in is developing multimodal imaging systems that allow the gathering of information from multiple techniques from a single specimen. An example of this is the recent combination of an optical nanoscope and atomic force microscope built on a customized Nikon platform.
Another goal is to implement faster techniques for super-resolution imaging through both software and hardware approaches. This might be through novel combinations of existing hardware, or by improved computational analysis of data during acquisition. An example of this would be being using on-the-fly data analysis during single molecule localization techniques to be able to stop acquisition once sufficient images have been acquired.
One of our biggest challenges is to be able to bring these high resolution imaging techniques to thick, highly scattering samples, tissues, organs, and ultimately in vivo. Some of the best candidates for this are methods based on multi-photon fluorescence excitation. As this is less efficient than single-photon excitation it means we require systems with very high optical performance.
What applications do you use the Nikon systems for?
We work closely with several teams regarding applications for the newly developed systems. We have a particular focus on molecular oncology (chromatin and adhesion mechanisms), the production of insulin in beta cells, and neuroscience (nerve degeneration, Alzheimer’s disease, brain mapping, neuronal network signaling).
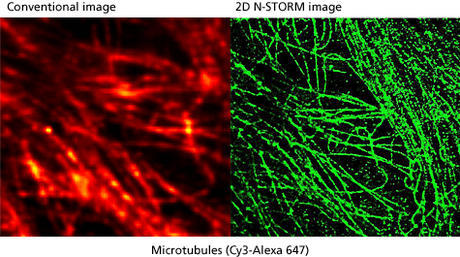
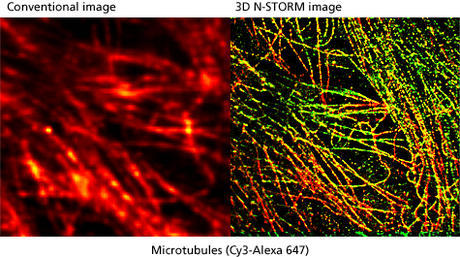
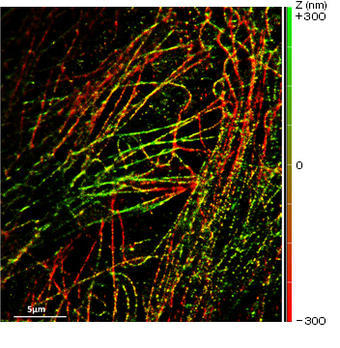
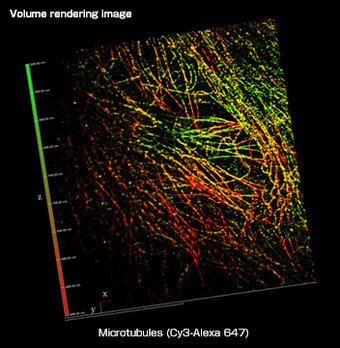
What was the deciding factor for choosing Nikon systems?
One of the main factors was Nikon’s reputation and experience in confocal laser scanning microscopy, which started with the PCM2000. Nikon systems are very efficient in terms of signal-to-noise ratio. When working at the nanoscale this becomes a critical factor.
Most importantly, the assistance given by Nikon is superb, with well qualified personnel and fast responses.
What was the performance specification that you valued most in selecting these systems?
The modularity and flexibility of the systems was very attractive as the basis for a platform for development. Nikon software is flexible and user friendly—Nikon has created some of the best software I’ve ever used. The Perfect Focus System is also very robust and useful for our time-lapse systems.
We are excited to explore the growing possibilities of intelligent microscope systems, such as CLEM (Controlled Light Exposure Microscopy).
What do you value about this Nikon system after having worked with it?
We have at our disposal a new-generation confocal system, which has been overbooked throughout the last year, and it has been up and running 100 percent of the time.
How has this system helped your research?
The systems have allowed us to make affordable measurements with high reproducibility. The systems are really high-value work horses for nanobiotechnology and neuroscience labs.

