- fr Change Region
- Global Site

Nikon BioImaging Labratories
Europe
Applications
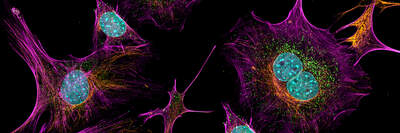
2D Imaging
We provide imaging services for a variety of 2D model systems. These services include a broad portfolio of assays, live and fixed cell imaging on slide coverslips, dishes and multi-well plates, epifluorescence or confocal imaging modalities, and intelligent image acquisition and AI-supported analysis. Our broad portfolio of assays captures our strength, and are highly customizable to client-specific needs.
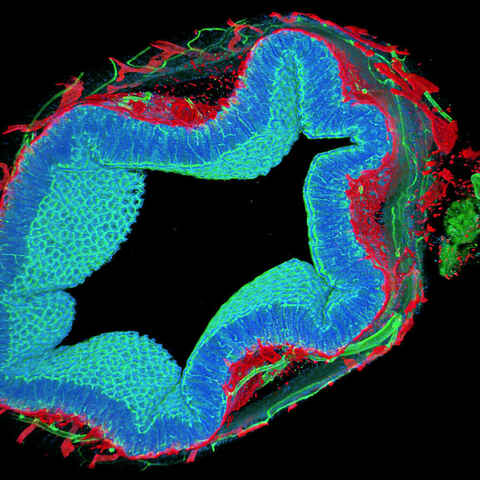
3D Imaging
We provide imaging services for a variety of 3D model systems. These services include a broad portfolio of assays, live and fixed cell imaging, time-lapse imaging, whole organism imaging, epifluorescence or confocal imaging modalities, intelligent image acquisition, and AI-supported analysis. Our broad portfolio of assays captures our strength, and are highly customizable to client-specific needs.
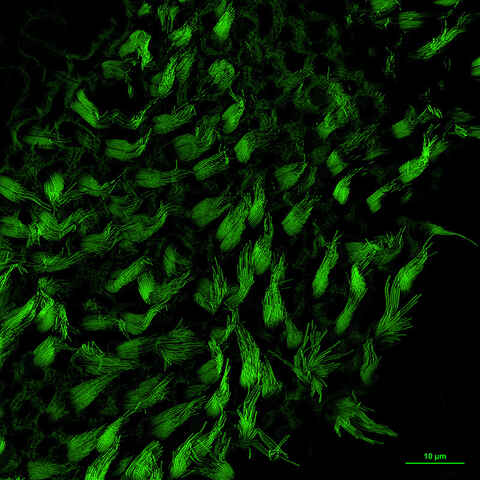
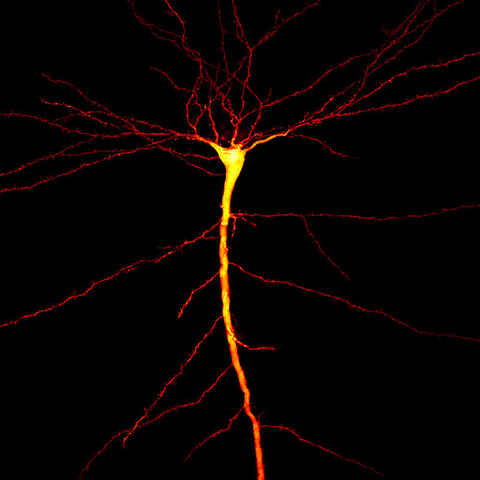
Super-Resolution
Unlock the power of ultra-detailed cellular imaging with super-resolution technology. Achieve unmatched clarity, ultra-low noise, and heightened sensitivity—ideal for revealing fine biological structures that standard microscopy can’t resolve.
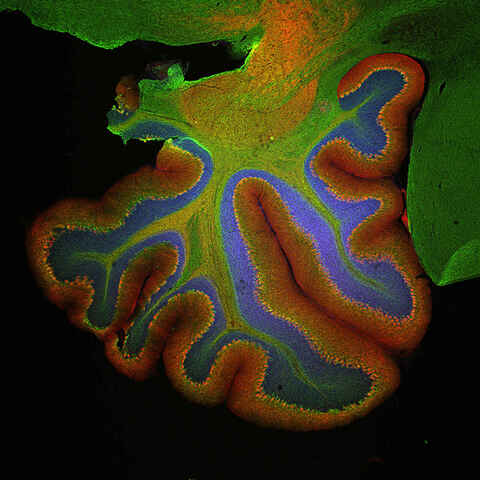
Whole Slide Scanning
We provide whole slide imaging services, brightfield or fluorescence, of fixed specimens for histology, pathology, gene expression, and spatial biology studies.
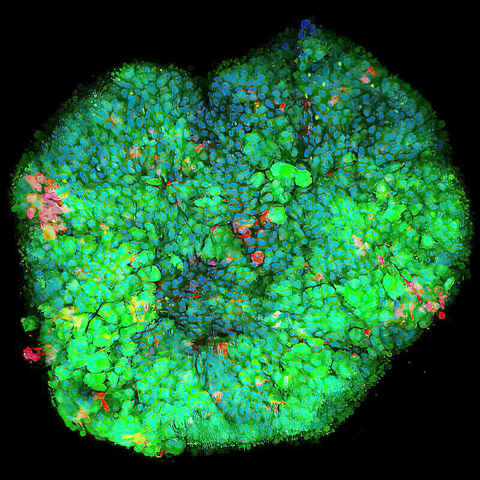
2D or 3D Toxicology, Uptake, and Viability Studies
This versatile service module provides customized bioimaging services for the purposes of studying cellular health and activity in 2D or 3D cultures. Possible experiments range from basic live/dead assays to organelle characterization in 3D spheroids.
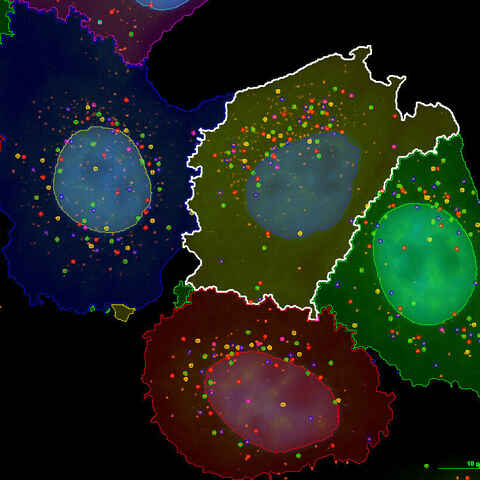
mRNA Quantification (smFISH) for Gene Expression Analysis
Single molecule fluorescence in situ hybridization (smFISH), known for its ability to quantify individual RNA molecules, is widely used to characterize spatio-temporal patterns of gene expression in single cells. At NBIL we harness this technique to provide RNA quantification services for research applications such as gene expression analysis in response to drug candidates, spatial biology studies, and more.
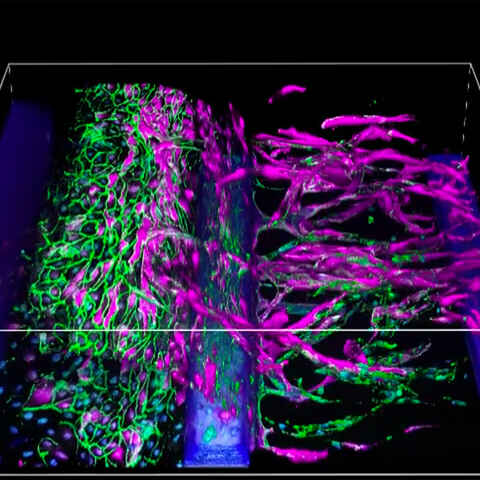
Organ-on-chip Applications
Organ-on-chip models enable the study of multicellular and tissue-level behavior in cells cultured on a scaffold that promotes organ-like conditions and morphology. These platforms are useful for providing a more physiological context to basic research and drug discovery experiments.
We collaborate with manufacturers such as Nortis, TissUse, and Mimetas. Customer-provided devices are welcome.
Automated Solutions for High-Content Imaging Applications
Generate consistent, high-quality data with Nikon’s turnkey automated microscopy and image analysis solutions.
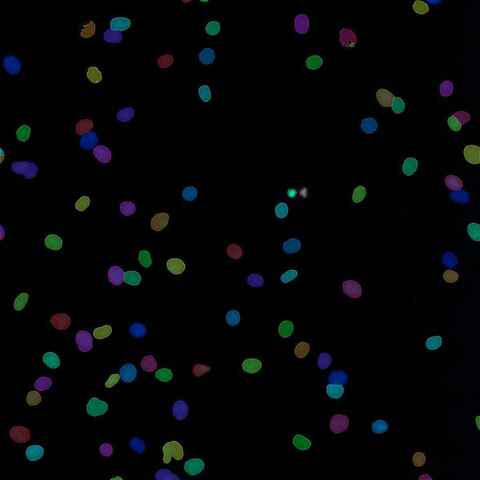
Custom Image Processing and Analysis
Image processing has a wide variety of uses and is an integral part of most microscopy experiments, from clarifying low-signal images to preparing images for robust segmentation and quantification. Custom analysis workflows are used for applications from gathering single-cell statistics to training AI networks for detection of customer-defined features.
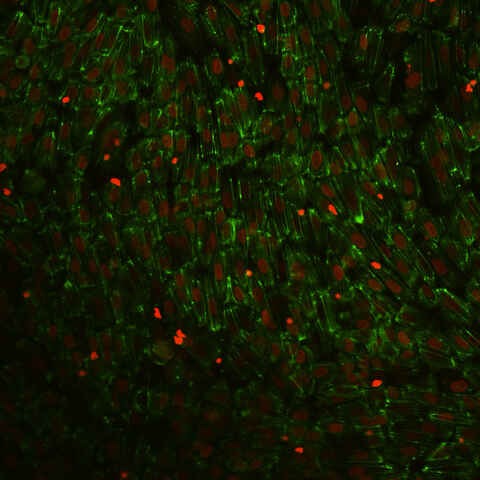
Transendothelial Migration
Endothelial cells (EC) line the lumen of blood and lymphatic vessels. Here these cells form a barrier and control tissue entry of cells and substances. The control over the entry of immune cells is mediated by a process termed transendothelial migration (TEM). Upon local inflammation endothelial cells attract and activate passing immune cells using chemokines and adhesion molecules. This activates immune cells and induces TEM, to alleviate inflammation. The TEM process in for example the invasion of tumor tissue by immune cells stands central to modern therapies like adoptive transfer of therapeutic T-cells or natural killer (NK) cells.
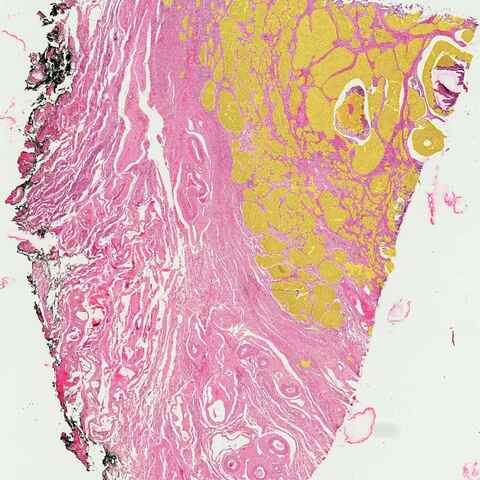
Histological Image Segmentation
Histological image analysis is a crucial aspect of medical research and diagnostics. It involves the microscopic examination of tissue samples that have been stained to highlight different cellular components and structures. This analysis allows pathologists to diagnose diseases, understand cellular interactions, and conduct detailed anatomical studies. The accuracy of histological analysis is vital, as it directly impacts clinical decisions and the understanding of complex biological processes.
